About
Harms provide important context for healthcare practitioners about the benefit-risk ratio of interventions. To improve transparency and credibility in the published results from randomized trials, the reporting of harms associated with an intervention needs to be explicit regarding what is patient-important, which may be different from that reported by clinicians submitting adverse event reports. Following the concerns about inadequate reporting of harm outcomes in randomized trials and systematic reviews the Outcome Measures in Rheumatology (OMERACT) Safety Working Group is advancing the work to identify additional harm aspects for assessment in rheumatology trials.
Current Stage in the OMERACT Process
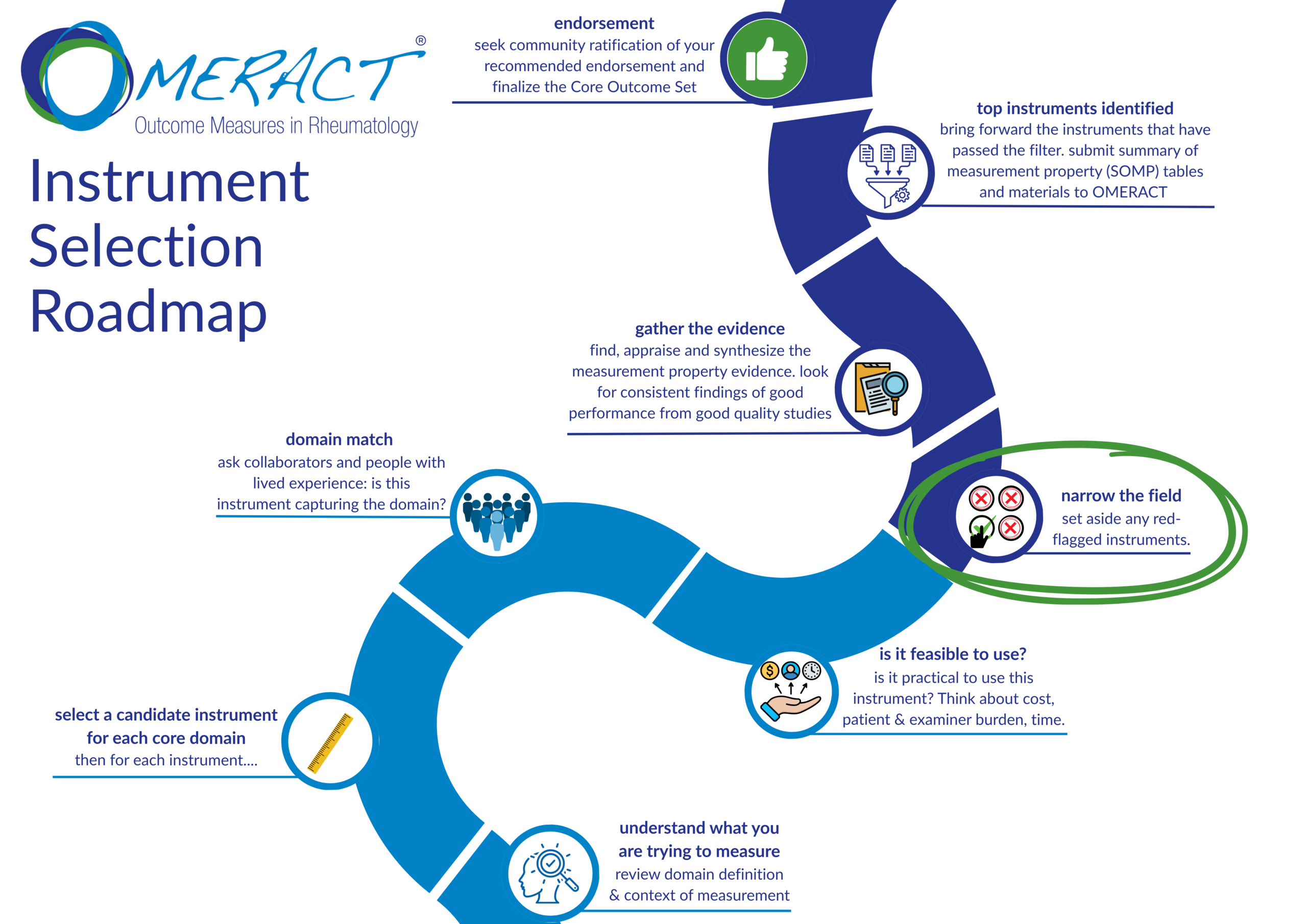
The Safety Working Group is developing and validating patient-reported tools to assess treatment harms in trials. Multiple projects, including single- and multi-item measures, are under review.
Latest Update
OMERACT 2025 Conference Highlights
Lightning Talk: In a rapid-fire session, our team shared an upcoming teaser of the Special Interest Group (SIG) session taking place during the conference.
👉 Watch the lightning talk here
Closing Plenary Talk: We were also featured in the Closing Plenary, where we highlighted the activities of the (SIG).
👉 Watch the presentation here

Recent Publications
- Berthelsen DB, Nielsen SM, Rasmussen MU, Voshaar M, Richards P, Bartlett SJ, Hazlewood GS, Shea BJ, Tugwell P, Ellingsen T, Jørgensen TS, Kristensen S, Simon LS, Christensen R, Flurey CA. 'I couldn't carry on taking a drug like that': a qualitative study of patient perspectives on side effects from rheumatology drugs. Rheumatology (Oxford). 2025 Mar 1;64(3):994-1002. doi: 10.1093/rheumatology/keae223. PMID: 38613847.
- Berthelsen, Dorthe B., et al. “Stakeholder Endorsement Advancing the Implementation of a Patient-Reported Domain for Harms in Rheumatology Clinical Trials: Outcome of the OMERACT Safety Working Group.” Seminars in Arthritis and Rheumatism, vol. 63, 152288, 2023, https://doi.org/10.1016/j.semarthrit.2023.152288.
- Berthelsen, Dorthe B., et al. “Harms Reported by Patients in Rheumatology Drug Trials: A Systematic Review of Randomized Trials in the Cochrane Library from an OMERACT Working Group.” Seminars in Arthritis and Rheumatism, vol. 51, no. 3, 2021, pp. 607–17, https://doi.org/10.1016/j.semarthrit.2020.09.023.
- Andersen, Kathleen M., et al. “Improving Benefit-Harm Assessment of Therapies from the Patient Perspective: OMERACT Premeeting Toward Consensus on Core Sets for Randomized Controlled Trials.” Journal of Rheumatology, vol. 46, no. 8, 2019, pp. 1053–58, https://doi.org/10.3899/jrheum.181123.
- Klokker, Louise, et al. “Identifying Possible Outcome Domains from Existing Outcome Measures to Inform an OMERACT Core Domain Set for Safety in Rheumatology Trials.” Journal of Rheumatology, vol. 46, no. 9, 2019, pp. 1173–78, https://doi.org/10.3899/jrheum.190196.
- Andersen, Kathleen M., et al. “Patient Perspectives on DMARD Safety Concerns in Rheumatology Trials: Results from Inflammatory Arthritis Patient Focus Groups and OMERACT Attendees Discussion.” Journal of Rheumatology, vol. 46, no. 9, 2019, pp. 1168–72, https://doi.org/10.3899/jrheum.181185.
- Klokker, Louise, et al. “Developing an OMERACT Core Outcome Set for Assessing Safety Components in Rheumatology Trials: The OMERACT Safety Working Group.” Journal of Rheumatology, vol. 44, no. 12, 2017, pp. 1916–19, https://doi.org/10.3899/jrheum.161105.
