About the Psoriatic Arthritis Working Group
Psoriatic arthritis (PsA) is an inflammatory musculoskeletal disease occurring in 7–42% of patients with psoriasis. Arthritis, enthesitis, dactylitis, spondylitis, and skin disease result in pain, stiffness, reduced mobility, impairment in physical function, and subsequent disability. PsA is now recognized as a disease that can be serious and progressive despite treatment, resulting in significant physical, psychological, functional, and social impairment.
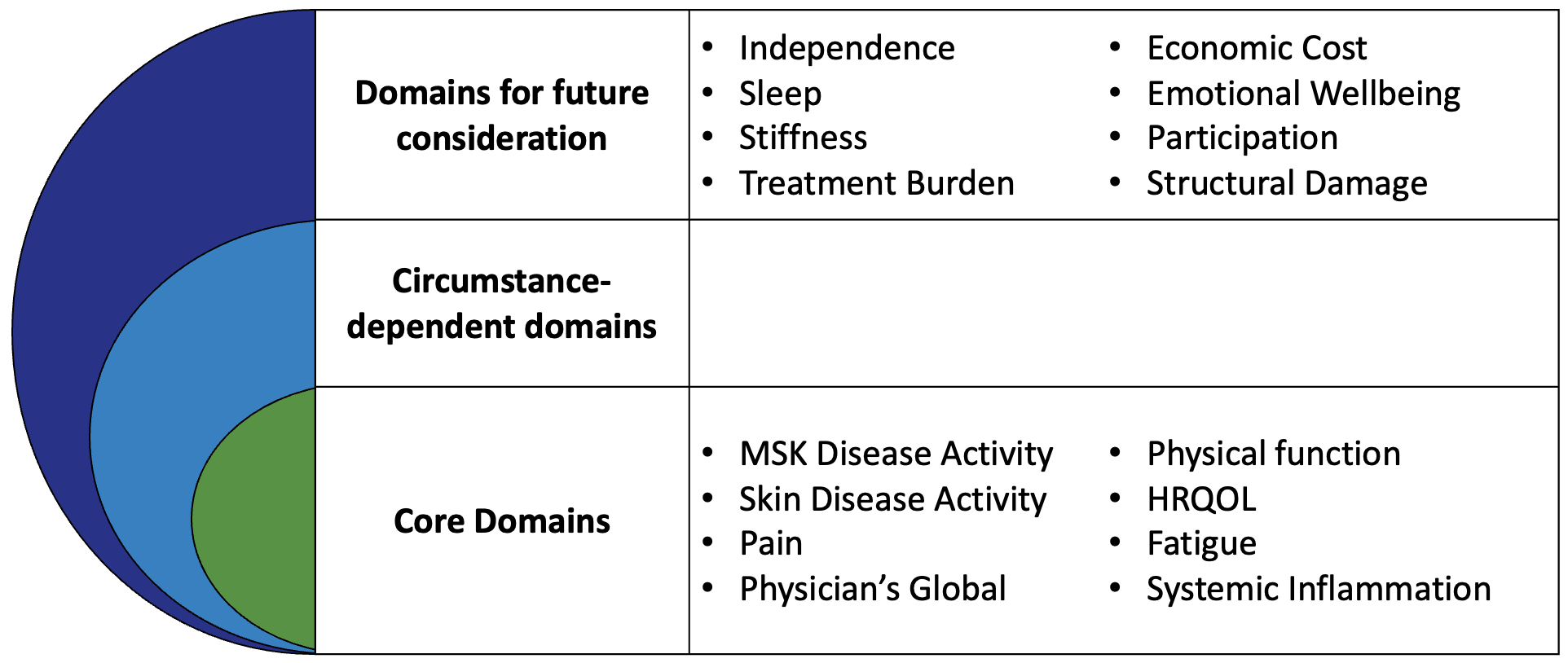
The updated PsA Core Domain Set was endorsed at OMERACT 2016. Next steps for the PsA working group include evaluation of PsA outcome measures and development of a PsA Core Outcome Measurement Set

Dafna Gladman
Co-Chair

Katy Leung
Co-Chair

Ana-Maria Orbai
Co-Chair

William Tillett
Co-Chair

Niti Goel
Patient Research Partner
OMERACT Endorsed Core Domain Set for Psoriatic Arthritis
Working Group Publications
OMERACT Filter 2.1 instrument selection for physical function domain in psoriatic arthritis: Provisional endorsement for HAQ-DI and SF-36 PF
Endorsement of the 66/68 Joint Count for the Measurement of Musculoskeletal Disease Activity: OMERACT 2018 Psoriatic Arthritis Workshop Report
PsAID12 Provisionally Endorsed at OMERACT 2018 as Core Outcome Measure to Assess Psoriatic Arthritis-specific Health-related Quality of Life in Clinical Trials
Updating the Psoriatic Arthritis (PsA) Core Domain Set: A Report from the PsA Workshop at OMERACT 2016
Enhanced patient involvement and the need to revise the core set- report from the Psoriatic Arthritis working group at OMERACT 2014
Development of a Disease Activity and Responder Index for PsA — Report of the PsA Module at OMERACT 11
Development of a Disease Severity and Responder Index for Psoriatic Arthritis (PsA) — Report of the OMERACT 10 PsA Special Interest Group
Consensus on a Core Set of Domains for PsA
Outcome Measures in PsA
Outcome Measures in Psoriatic Arthritis
Working Group Members:
Adenike Adebajo
Adewale Adebajo
Adham Khalil
Adrien Nzeusseu Toukap
Alireza Meysami
Amaury Brianez
Ana-Maria Orbai
Andre Ribeiro
Andrea Falzon
Anetta Chrzanowska
Anupam Wakhlu
Ashish Mathew
Aya Akmal Amin
Beverley Shea
Boo Edgar
Carlos Guillen Astete
Christine Lindsay
Chu Hieu chi
Clifton (Bing) Bingham
Dafna Gladman
Denis O'Sullivan
Denise Bury
Dennis Neuen
Desiree van der Heijde
Edward Henstridge
Elena Garcia Guijarro
Eman Fekry
Esther Francisca Vicente-Rabaneda
Fatma Ali
Fernando Pimentel-Santos
Fernando Saraiva
Florian Naye
Francesco Caso
Frederic Lavie
Gary Choque
Gehad Maghraby
George Bruyn
Ghaydaa Dabie
Gizem Ayan
Gordon Hendry
Heidi Bertheussen
Ilfita Sahbudin
Ingrid Moller Parera
Ingrid Steinkoenig
Jacob Brauner
Jane Hayes
Javier Rios
Jean-Noel Talabardon
Jeffrey Chau
Jo Merola
Joao Dias
Jose Alexandre Mendona
Kapil Garg
Karen Natali Saravia Chocobar
Kate Smith
Kategia Binion
Kenta Misaki
Khaled Abdelsalam Abdelgalil
Khaled Saadaoui
Khalid Alnaqbi
Kristina Duffin
Kyung Min Ko
Lara Fallon
Latika Gupta
Laura Coates
Laure Gossec
Lays De Souza
Lihi Eder
Liliia Shvets
Lizeth Chaparro Del Portillo
Maarten de Wit
Madusha Menu Cristeen Jayasinghe
Maria Antonietta d'Agostino
Maria Queiroz
Maria Sofia Ciliento
Maria Stoenoiu
Maria Teresa Romero de Albrecht
Maria-Sole Chimenti
Maxine Isbel
Michel Yazbek
Mikael Boesen
Minh Vu
Minna Kohler
Mirco Magnani
Mona Hamdy
Monica Vieth
Mustafa Serdar Cantez
Nino Tsiskarishvili
Niti Goel
Nouran Abaza
Oliver Fitzgerald
Oliver Kramer
Olympia Papakonstantinou
Panagiotis Ermeidis
Peter Nash
Peter Tugwell
Philip Conaghan
Philip Mease
Philippe Carron
Pil Hojgaard
Raouf Hajji
Raquel Granados
Richard Holland
Robin Christensen
Russka Shumnalieva
Salih Ozgocmen
Sasikala Bheemireddy
Sevdalina Lambova
Shawna Grosskleg
Simay Akdemir
Simeon Grazio
Simon Stones
Soumya Chakravarty
Tamer Gheita
Tania Elena Gudu
Teresa Clavaguera
Terry Lowdon
Tommy Kok Annfeldt
Tuncay Duruoz
Valeriia Riazanova
Vibeke Strand
Walter Maksymowych
Will Taylor
Willemina Campbell
Ying Ying (Katy) Leung

