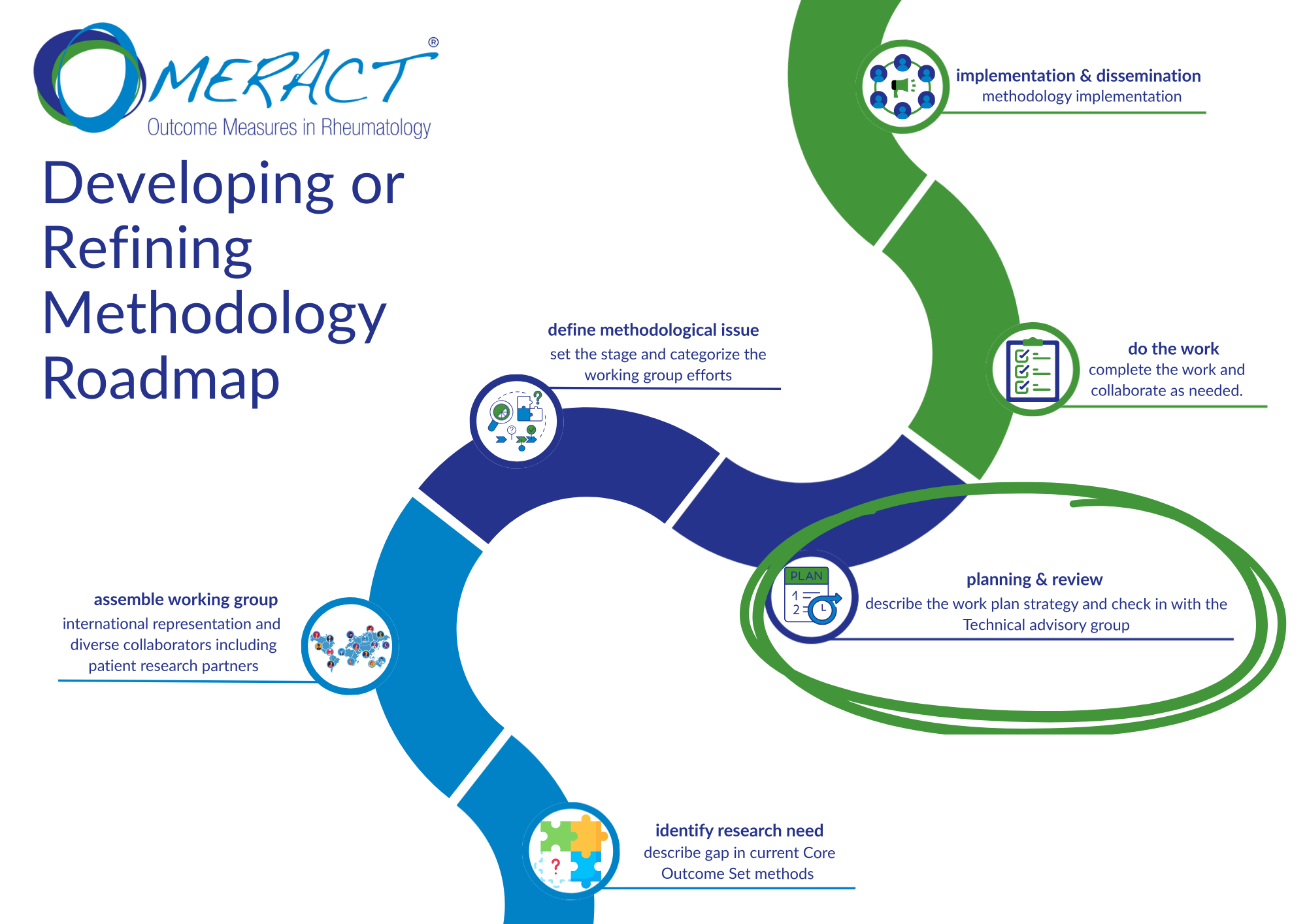
About
Disease prevention and risk-stage research are emerging rapidly across immune-mediated rheumatic diseases. In rheumatoid arthritis (RA), a well-defined pre-clinical phase exists in which individuals develop anticitrullinated protein antibodies (ACPA) prior to the onset of clinically apparent disease. Increasing evidence suggests that intervention during this phase may delay or prevent disease onset or reduce disease severity.
Clinical trials targeting at-risk populations are expanding, including both pharmacological and lifestyle interventions. However, there is currently no consensus on which outcomes should be measured in prevention trials, and existing studies demonstrate substantial heterogeneity in outcome selection.
While a Core Domain Set (CDS) exists for established RA, prevention trials involve fundamentally different populations and objectives. Individuals at risk of disease represent a distinct clinical and conceptual state, and outcomes relevant to established disease may not adequately capture what matters in prevention contexts.
This Working Group aims to address this gap by developing guidance on developing a Core Outcome Set for prevention and risk-stage trials in rheumatic diseases, using RA as an initial exemplar.