About
Gout is the most common form of inflammatory arthritis and is caused by deposition of monosodium urate (MSU) crystals in and around joints, which provokes an intense inflammatory response. Gout manifests as episodes of intense joint pain, swelling, and functional disability that may lead to persistent joint symptoms and structural joint damage.
In our pursuit of ensuring that research aligns with the needs and interests of those most affected by gout, our team has diligently worked on the identification and consensus of the most crucial outcomes to be measured in all clinical trials and observational studies of gout.
Our work so far has incorporated key elements of the OMERACT Framework, including a thorough literature review, Delphi surveys, and patient involvement at all stages of the research process. We have developed a Core Domain Set that reflects not only the clinical aspects of gout but also the lived experience of patients.
Current Stage in the OMERACT Process
Building on the progess we've made, our next focus is to provide explicit guidance on the measurement methods and reporting needs for the core outcome set.
Latest Update
The Gout Working Group is developing clear guidance on how key outcomes should be measured and reported. Their work supports consistency and comparability across clinical trials.

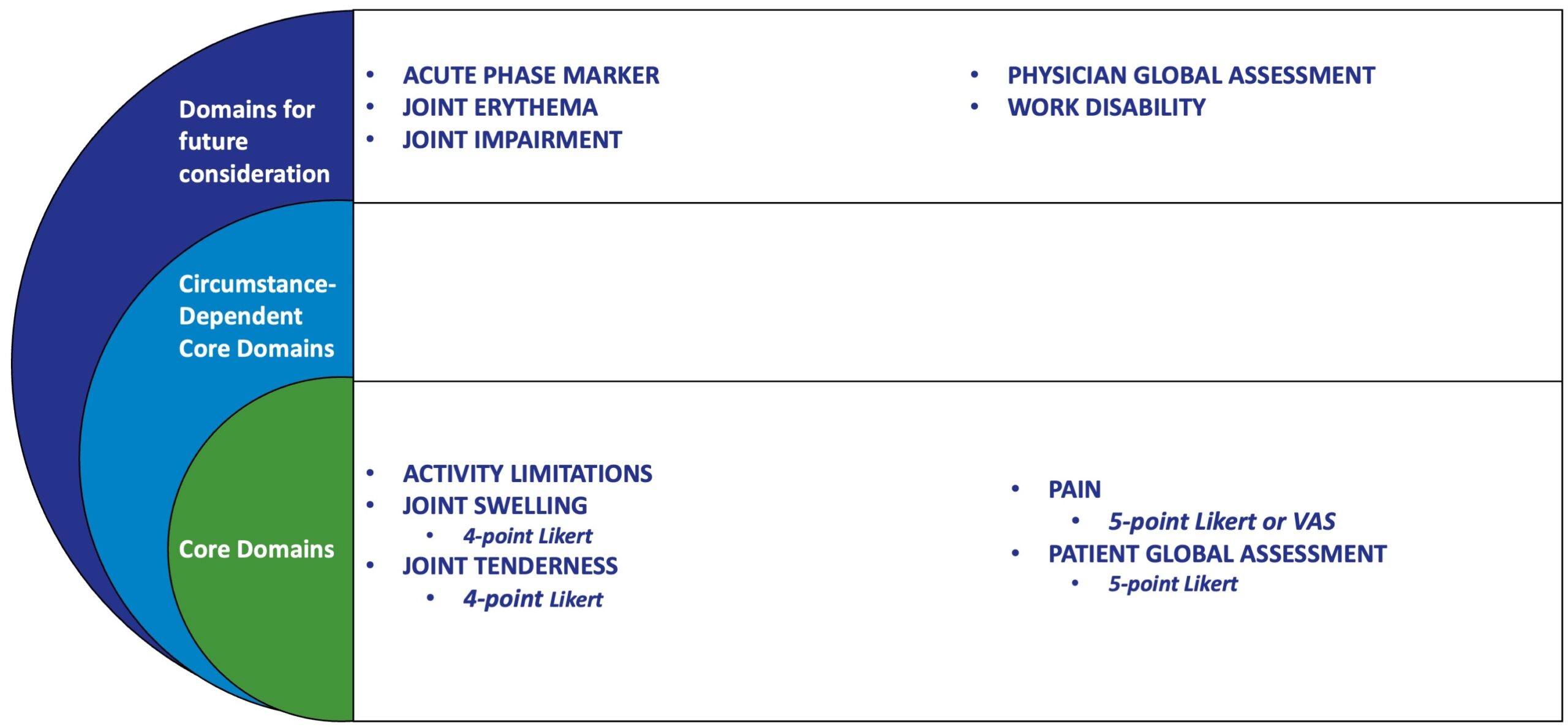
ACUTE GOUT
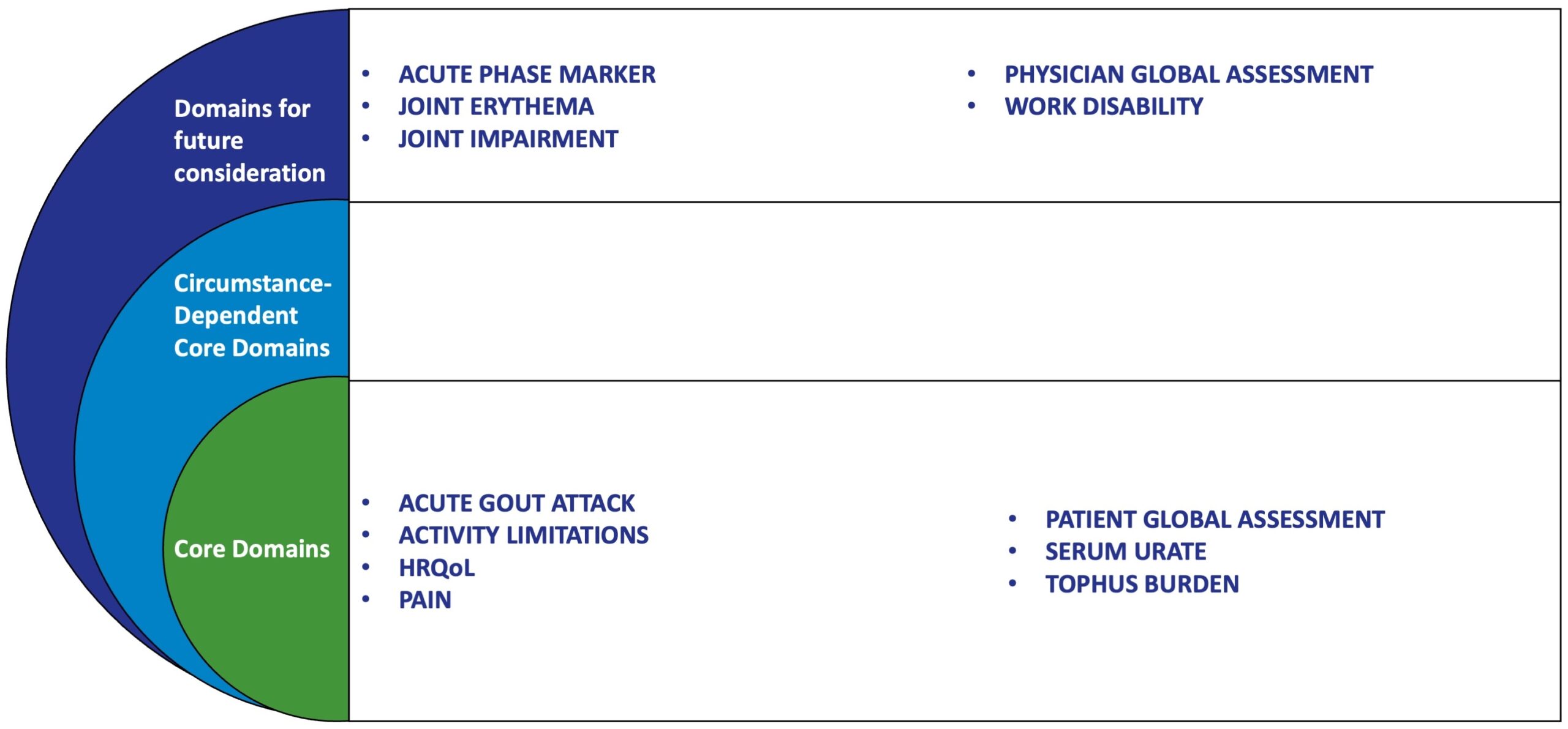
CHRONIC GOUT
Recent Publications
Morillon, Melanie B., et al. “Outcome Reporting in Randomized Trials in Gout: A Systematic Scoping Review from the OMERACT Gout Working Group Assessing the Uptake of the Core Outcome Set.” Seminars in Arthritis and Rheumatism, vol. 60, 152191, 2023, https://doi.org/10.1016/j.semarthrit.2023.152191.
Morillon, Melanie Birger, et al. “Serum Urate as a Proposed Surrogate Outcome Measure in Gout Trials: From the OMERACT Working Group.” Seminars in Arthritis and Rheumatism, vol. 51, no. 6, 2021, pp. 1378–85, https://doi.org/10.1016/j.semarthrit.2021.11.004.
- Singh, Jasvinder A., et al. “OMERACT Endorsement of Measures of Outcome for Studies of Acute Gout.” Journal of Rheumatology, vol. 41, no. 3, 2014, pp. 569–73, https://doi.org/10.3899/jrheum.131246.





